If you have anyquestions
-
Call Now
-
Any Questions medrainbow.info@gmail.com

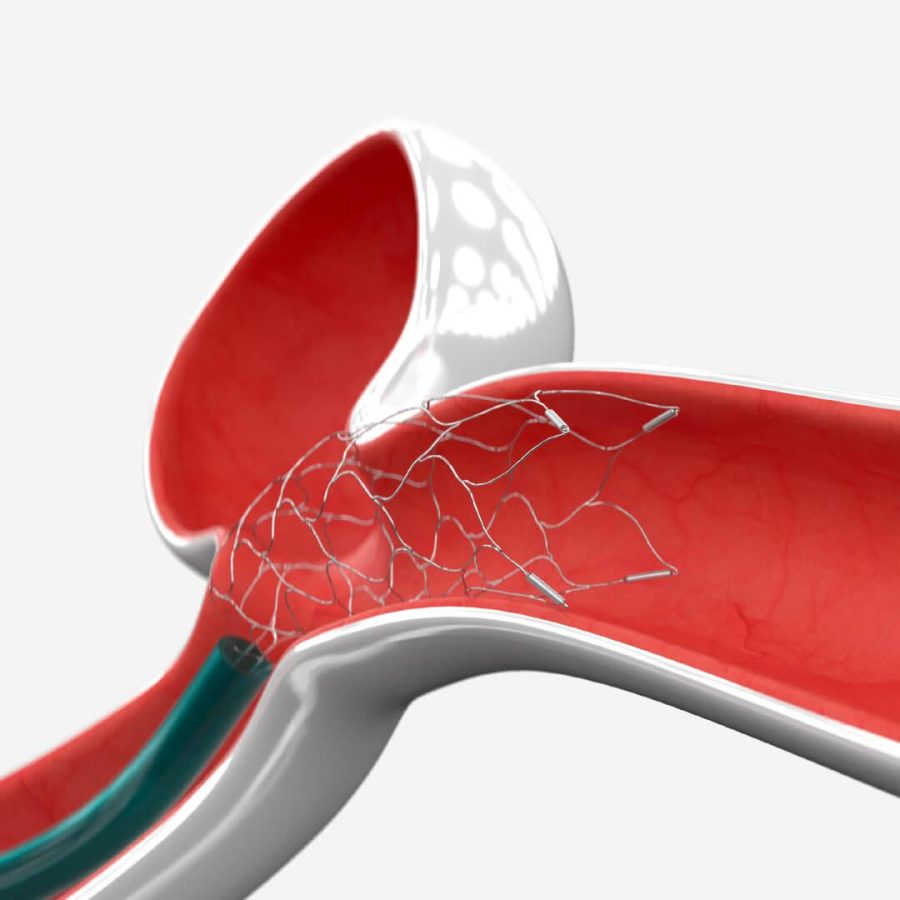
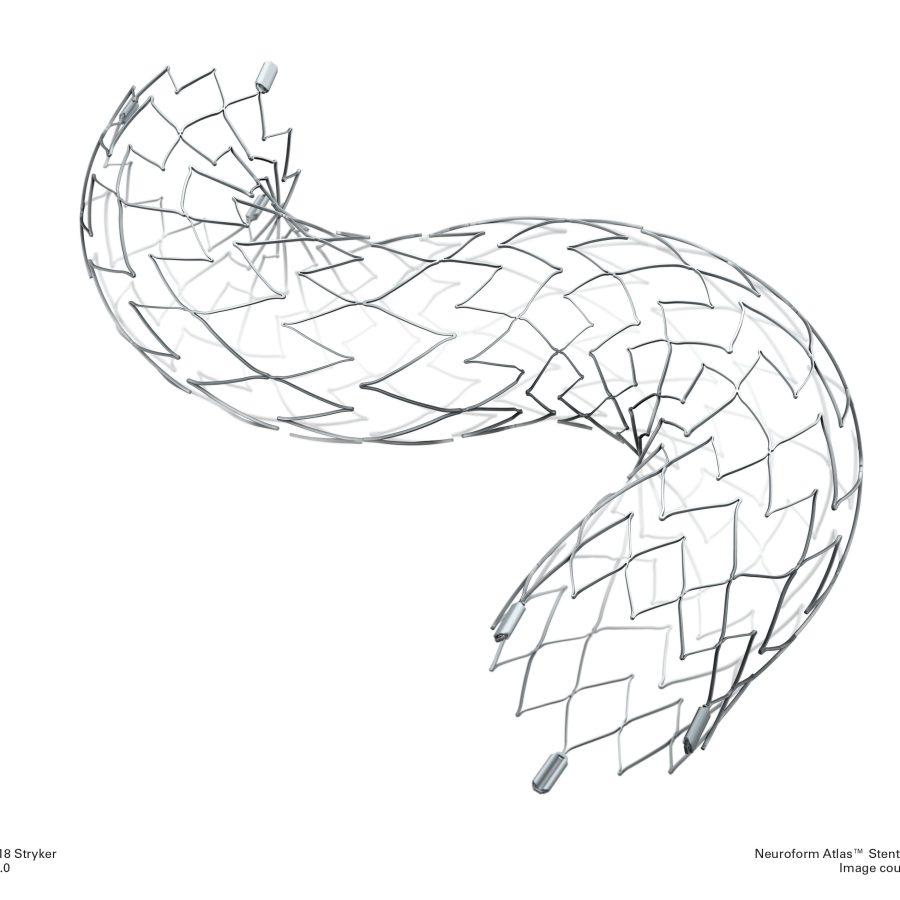
ENTERPRISE Vascular Reconstruction Device and Deliver血管重建装置和传送系统
| Brand | CERENOVUS |
|---|---|
| Manufacturer | Medos International SARL迈道国际有限公司 |
| Production Address | 340 Lake Hazeltine Dr., Chaska, MN 55318, USA |
| Intended Use | This product is used together with the embolic device for the treatment of intracranial aneurysms.该产品与治疗颅内动脉瘤的栓塞装置一起使用。 |
| References | ENC452212,ENC452812 …… |
| Manufacturer’s Website | https://www.jnjmedtech.com/en-US/product/enterprise-2-vascular-reconstruction-device |
| Product Brochure | |
| Registration Certificate | 国械注进20173130352 |
This product consists of a stent and a conveying system, where the conveying system includes a conveying wire and an inserter. The bracket is pre-installed on the conveying wire inside the inserter. The bracket is made of nickel-titanium alloy material and designed in a closed form. There are four tantalum markers at each end of the bracket, and the outer surface of the bracket is coated with a layer of polymer. The conveying wire is composed of a nickel-titanium alloy wire core with X-ray opaque markers. It is sterilized with ethylene oxide, for one-time use, and has a shelf life of two years.
该产品由支架和传送系统组成,传送系统包括一条传送导丝和一个导入器。支架预先安装在导入器内部的传送导丝上。支架由镍钛合金材料制成,采用闭合式设计。支架每一端有四个钽标记带,支架外表面涂有一层聚合物。传送导丝由带有不透X线标记的镍钛锘导丝芯构成。环氧乙烷灭菌,一次性使用,有效期二年。
Product List
| REF | GTIN | Classification Code | Description |
|---|---|---|---|
| ENC451400 | 10886704071464 | 13-06-06 | 血管重建装置和传送系统 ENC451400 |
| ENC451412 | 10886704043973 | 13-06-06 | 血管重建装置和传送系统 ENC451412 |
| ENC452200 | 10886704071471 | 13-06-06 | 血管重建装置和传送系统 ENC452200 |
| ENC452212 | 10886704043980 | 13-06-06 | 血管重建装置和传送系统 ENC452212 |
| ENC452800 | 10886704071488 | 13-06-06 | 血管重建装置和传送系统 ENC452800 |
| ENC452812 | 10886704043997 | 13-06-06 | 血管重建装置和传送系统 ENC452812 |
| ENC453700 | 10886704071495 | 13-06-06 | 血管重建装置和传送系统 ENC453700 |
| ENC453712 | 10886704044000 | 13-06-06 | 血管重建装置和传送系统 ENC453712 |
| Risk-Based Classification风险管理类别 | |
|---|---|
| Product Status产品状态 | Active |





Reviews
There are no reviews yet.